
Section Navigation
9. Learning from Others
9.1
Introduction: Grouping by Business Models
:Cautionary Tales
9.2
A Start
9.3 Coins International
9.4 Fine Art Ceramics
9.5 Halberd Engineering
9.6
Ipswich Seeds
9.7 Seascape e-Art
9.8 Whisky Galore
:Case
Studies
9.9 Amazon
9.10
Andhra Pradesh
9.11 Apple iPod
9.12 Aurora Health Care
9.13
Cisco
9.14 Commerce Bancorp
9.15 Craigslist
9.16
Dell
9.17 Early Dotcom
Failures
9.18 Easy Diagnosis
9.19 eBay
9.20
Eneco
9.21 Fiat
9.22
GlaxoSmithKline
9.23 Google ads
9.24 Google services
9.25
Intel
9.26 Liquidation
9.27
Lotus
9.28 Lulu
9.29
Netflix
9.30 Nespresso
9.31
Netscape
9.32 Nitendo wii
9.33 Open Table
9.34
PayPal
9.35 Procter & Gamble
9.36 SIS Datenverarbeitung
9.37 Skype
9.38
Tesco
9.39 Twitter
9.40
Wal-mart
9.41 Zappos
9.42
Zipcar
 9.22
GlaxoSmithKline plc
9.22
GlaxoSmithKline plc
GlaxoSmithKline, a British pharmaceutical, biological, and healthcare company, is the world's fourth largest pharmaceutical company (by revenue) after Johnson & Johnson, Pfizer, and Roche. GSK was formed in 2000 by the merger of GlaxoWellcome plc with SmithKline Beecham plc, and today markets a wide range of pharmaceutical products (anti-infectives, vaccines, central nervous system, respiratory, gastro-intestinal/metabolic and oncology drugs), healthcare products, nutritional drinks, and over-the-counter medicines. The companies have grown organically and by a complex series of mergers and takeovers, but Glaxo started as a baby food manufacturer in 1904, and Beechams with laxative pills in 1843. {1} {2}
Big Pharma
Drugs are the mainstay of modern medicine, and the pharmaceutical industry employs legions of entirely reputable scientists to improve the range and effectiveness of its many thousand of products. {2} The industry has an unattractive public image, {26} however, and the list of its alleged sins is a long one. Big pharma is accused of exerting excessive lobbying power, {3} of a 'revolving door' association with the FDA, {4} of colluding with psychiatrist to invent disorders, {5} {7} of keeping prices unreasonably high by preventing Medicare buying at bulk discounts, {6} of preventing US citizens from purchasing at Canadian prices, {6} of preventing licenses lapsing by insignificant patent modification, {6} of accepting heavy fines for malpractice as part of business, {9} {25} of promoting favorable articles in academic journals, {13} of buying influence among doctors, hospitals and research laboratories {14} and of confusing the public generally. {8} {15} {27-8} Supporters of the FDA do not deny its close association with pharmaceutical companies, {11} nor its war on natural health food and product claims, {12} but argue that the authority does its best with a near-impossible agenda. {10} {11}
Zantac
Glaxo overcame the market dominance of Tagamet, the leading ulcer drug developed by SmithKline Beecham, with their 1981 Zantac drug. Though the FDA rated Zantac as making 'little or no' contribution to existing drug therapies, Glaxo promoted Zantac to the number one pharmaceutic product in the world by:
1. Quickly
introducing the drug worldwide.
2. Extensive partnerships with distributors.
3.
Articles in medical journals on the negative effects of Tagamet and potential for
Zantac.
4. Simplifying the dosage, from 4 to 2 pills a day.
5. Marketing as
'fast, simple and specific' (which doctors interpreted as 'faster, simpler and safe').
6. Pricing Zantac at a slight premium over Tagamet.
SmithKline did not properly defend its product, and Zantac achieved a 42% global market share, with sales amounting to US$1 billion by 1989. {16}
The example proved a turning point for Big Pharma, which cut back on research to make higher profits through advertising.
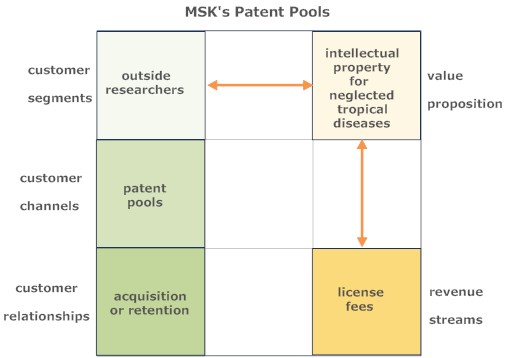
Patent Pools
Company image was probably one factor in GSK's decision to create a patents pool for neglected tropical diseases, aggregating intellectual property rights to avoid R&D advances being blocked by a single rights holder. These patents were not likely to lead to blockbuster drugs that make big profits for pharmaceutical companies, but could facilitate the development of drugs vital to the world's poorest countries. {17} The pool currently contains over 2,300 patents accessible to industry, research and funding agencies.{18} In business model terms, this represents an inside-out approach, monetizing unused internal assets, and was recognized as such. Knowledge Ecology International {19} point out that:
1. GSK were in reality seeking to
benefit from the new interest in Type III disease research.
2. More general and
disinterested patent pools are being developed by the priority review voucher (PRV),
by UNITAID and the new WHO Global Strategy And Plan Of Action On Public Health, Innovation
And Intellectual Property.
3. Philanthropy is simply another pharma-friendly
approach being developed to head off the reforms needed by the industry, particularly
in the developing world. (Others approaches include the Advanced Marketing Commitment
R&D subsidies, the Pogge/Hollis effort, and the Barton/Pfizer proposal to regulate
drug prices in middle income countries.)
Requip
Ropinirole (sold as Requip, Ropark or Adartrel) is a non-ergoline dopamine agonist manufactured by SGK and used in the treatment of Parkinson's disease and (latterly) restless legs syndrome (RLS). {20} Because Ropinirole's use for Parkinson's disease had already passed the toxicity and other tests that cause over 90% of drugs to fail during development, GSK were able to reposition Ropinirole for RLS, receiving FDA approval and so considerably extend its market. RLS was a relatively unknown disorder until SGK mounted an intensive marketing campaign, in stages:
1. USA: Prior to FDA approval (March 2003 to May 2005)
1. RLS awareness campaign: presentations at conferences and numerous articles in medical journals. Some appeared in the mainstream press.
2. USA: After FDA approval (from May 2005)
1. Multi-million dollar, direct to consumer, TV advertising: first creating interest in the condition, and then promoting Requip. Sales of Requip, which had been steady from its launch in 1997, grew rapidly: $156 million in 2005, $268 million in 2006, an expected $1,700 million in 2015.
3. Marketed as Adratrel in Europe (from June 2004)
1. Adratel had
been accepted in France (June 2004) and the UK (April 2005).
2. Lobbying of the
European Commission got general EMEA approval in September 2005 and Adratrel accepted
in Spain and the Netherlands (both in April 2006).
3. Acceptance was delayed
by tactics disliked in Europe, e.g. ads in medical journals and direct selling to
doctors. {20}
GSK used the best of marketing techniques: TV, web pages and CD presentational aids combining video, audio and graphical display tools.
Controversies
Opinion was split, between those (patients, some doctors) who thought GSK were to be commended for raising awareness of RLS, and those (mostly doctors) who thought the condition was over-hyped in prevalence and gravity. Professionals were generally unhappy to see a medical matter captured by commerce, accusing GSK of 'disease mongering' and placing RLS with other concocted maladies like erectile dysfunction, social anxiety disorder, etc. GSK donations to the Restless Leg Sufferers' Foundation and Sleeptionary (a website created by the National Sleep Foundation) were also criticized. Patients retorted that RLs was real enough to its sufferers, and that disorder trailblazing was far from new, Listerine having coined the condition 'halitosis' back in the 1930s. Advertisers were generally impressed by the effectiveness of the Requip campaign and its success in making RLS a household term. Awards were given to GSK for excellence in advertising, and other pharmaceutical companies took note of the approach and developed their own remedies for RLS. {20}
Current Threats
1. None of the pharmaceutical companies is at risk from US government regulation
while the FDA and lobbying industries continue to exert influence.
2. China,
India and other countries have shown less willingness to be bound by international
patents. {21}
3. Many pharmaceutical companies face aging patents and lack of
obvious replacements: profits are declining. {22} {23} {24}
Points to Note
1. Successful marketing campaigns need not be truthful.
2. Key partners can be vital, here Congress and the FDA.
 Questions
Questions
1. Is Big Pharma's public image as here portrayed either accurate or justified?
Would it matter anyway — i.e. does ethics enter into business, and can 'value
to customers' become simply 'perceived value'?
2. How did GlaxoSmithKline market
their Zantac drug? Why was the marketing important, and what lessons can be learned?
3. Describe GlaxoSmithKline's Patent Pool, and the business model that applies.
4. Give a marketer's view of the Ropinirole promotion.
5. Outline the difficulties
currently faced by the pharmaceutical industries, and suggest some remedies.
 Sources
and Further Reading
Sources
and Further Reading
1. GlaxoSmithKline. LegalView.
Summary of company and recent activities.2. Glaxo Holdings. Funding Universe. Straightforward, positive business account.
3. Industry Puts $44 Million into State Lobbying Campaigns push back against moves to cut prices, spending on medication by M. Asif Ismail. Public Integrity. April 2006.
4. Moody Is the New Bipolar. Alternet. November 2007.
5. Citizens Group Calls The Diagnostic And Statistical Manual of Mental Disorders a Fraud. PRLog. May 2010.
6. The Truth About the Drug Companies by Marcia Angell. CorpWatch July 2004.
7. Anatomy of an Epidemic: Magic Bullets, Psychiatric Drugs, and the Astonishing Rise of Mental Illness in America by Robert Whitaker. Broadway. August 2011.
8. The STAR*D Scandal Home Anatomy of an Epidemic by Ed Pigott. Mad in America. April 2011.
9. Big Pharma Absorbs $5 billion in fines! 'The cost of doing business!' Big Red Biotech Blog. February 2010.
10. Reputation and Power: Organizational Image and Pharmaceutical Regulation at the FDA by Daniel Carpenter. Princeton Univ. Press. April 2010. NYT Review.
11. Inside the FDA: The Business and Politics Behind the Drugs We Take and the Food We Eat by Fran Hawthorne. Wiley. February 2005.
12. FDA's scheme to outlaw nearly all nutritional supplements created after 1994 would destroy millions of jobs and devastate economy by Mike Adams. Natural News. July 2011.
13. Threats to science-based medicine: Big pharma pays a publisher to produce a fake journal by David Gorski. Science-Based Medicine. May 2009.
14. Let Them Eat Prozac by David Healey. NYU Press. October 2006. Let Them Eat Prozac Explores threats to public safety and academic freedom.
15. Big Pharma opts to publish funding of doctors by Andrew Jack. FT. August 2011.
16. Strategy from the Outside In by George S. Day and Christine Moorman. McGraw Hill. 2010. 183-4.
17. Business Model Generation: A Handbook for Visionaries, Game Changers, and Challengers by Alexander Osterwalder and Yves Pigneur. Wiley. 2010. 113.
18. Pool for Open Innovation. Neglected Tropical Diseases Pool. More than than 2,300 patents accessible to industry, research and funding agencies.
19. KEI reaction to GSK announcement on patent pool for neglected diseases. Knowledge Ecology International.
20. Glaxosmithkline's Marketing Strategy for Requip: A Case Study in Product Lifecycle Management. ImcrIndia. Detailed treatment with extensive references.
21. Protecting personalized medicine innovation in China and India: Are diagnostic methods patentable? NixonPeabody. May 2011.
22. Generics' new legal attack: Big Pharma's aging patents by Kit R. Roane. CNNMoney. March 2011.
23. Pharmas forced to put squeeze on R&D by Andrew Jack. FT. October 2011. Only Novartis has NPV of new drug approvals greater than R&D spend 2007-11.
24. Big Pharma Faces Grim Prognosis: Industry Fails to Find New Drugs to Replace Wonders Like Lipitor by Barbara Martinez and Jacob Goldstein. WSJ. December 2007.
25. GlaxoSmithKline to pay $3 billion in fines, the largest health care fraud settlement in U.S. history. NY Daily News. July 2012.
26. An Open Letter To The PhRMA CEO On Pharma's Image by John L. LaMattina. Forbes. April 2013.
27. A New Flu Season of Pain, Profit and Politics by Gary Null and Richard Gale. Global Research. November, 2013.
28. Bitter Pill: Why Medical Bills are Killing Us by Steven Brill. Time Magazine. February 2013.